Endoscopic stenting versus operative gastrojejunostomy for malignant gastric outlet obstruction-a systematic review and meta-analysis of randomized and non-randomized trials
Introduction
Gastric outlet obstruction (GOO) is a recognised complication of malignancies of the upper gastrointestinal (UGI) tract. The most common causes are pancreatic and gastric malignancies, with lymphomas, ampullary carcinomas, biliary tract cancers and metastases also contributing. In patients with pancreatic cancer, it is estimated that 15-20% of patients develop GOO (1). The majority of patients have locally advanced or metastatic cancer with dismal prognosis and median survival of only 3-6 months (2). The aim in palliating patients with malignant GOO is to re-establish an oral intake by restoring gastrointestinal continuity. This ultimately improves patients’ quality of life in the advanced stages of cancer. Traditionally, surgical gastrojejunostomy (GJ) has been the standard treatment approach for these patients. Although GJ relieves symptoms in almost all patients, the procedure is associated with morbidity of 10-16% and mortality of up to 7% (3-5). Also, post-operatively, most patients suffer delayed gastric emptying that is often associated with longer hospital stay (6). Although laparoscopic GJ has been introduced as a less invasive alternative to open GJ, the technique still carries substantial risk and is not widely available (7-10).
Endoscopic placement of self-expandable metal stents (SEMSs) has emerged as an alternative means for palliation of GOO. Multiple uncontrolled case-series studies have demonstrated SEMSs to be safe and effective with technical success of 90-100% and clinical success of 67-100% (11-17). Randomized trials have shown mixed results, with two trials favouring endoscopic SEMS (18,19) and one favouring surgical GJ (20). Therefore, it is currently unknown whether patients with GOO are best palliated with endoscopic SEMS placement or GJ. Also, SEMS are expensive and it is unclear whether their use is less costly when compared with surgical GJ. Although direct cost studies have shown that SEMS placement is less costly than surgery, the general applicability of the data is debatable given the small number of patients enrolled in each of these single-institution trials (7,21,22).
Hence we performed this meta-analysis to compare outcomes of endoscopic stenting (ES) with GJ. The primary goal of this study is to the compare the overall complication rate and effectiveness (ability to tolerate oral intake) of SEMS and GJ in patients with GOO. The secondary objective is to identify predictors of clinical outcomes [reintervention rate, length of hospital stay (LOHS), hospitalization charges, and complications].
Methods
Study protocol
We followed the Preferred Reporting Items for Systematic reviews and Meta-Analyses PRISMA guidelines where possible in performing our systematic review (23). We performed a systematic search through MEDLINE (from 1950), PubMed (from 1946), EMBASE (from 1949), Current Contents Connect (from 1998), Cochrane library, Google scholar, Science Direct, and Web of Science to January 2013. The search terms included “gastric outlet, gastroduodenal or duodenal obstruction’’, ‘‘gastrojejunostomy, gastroenterostomy or surgical bypass’’, and ‘‘endoscopic and stent”, which were searched as text word and as exploded medical subject headings where possible. No language restrictions were used in either the search or study selection. The reference lists of relevant articles were also searched for appropriate studies. A search for unpublished literature was not performed.
Study selection
We included studies that met the following inclusion criteria:
• Studies identifying the population of patients with GOO who underwent GJ or SEMS.
Data extraction
We performed the data extraction using a standardized data extraction form, collecting information on the publication year, study design, number of cases, total sample size, population type, country, continent, mean age and clinical data. The event rate and confidence intervals (CI) were calculated.
Statistical analysis
Pooled event rate and 95% CI were calculated using a random effects model (24). We tested heterogeneity with Cochran’s Q statistic, with P<0.10 indicating heterogeneity, and quantified the degree of heterogeneity using the I2 statistic, which represents the percentage of the total variability across studies which is due to heterogeneity. I2 values of 25%, 50% and 75% corresponded to low, moderate and high degrees of heterogeneity respectively (25). The quantified publication bias using the Egger’s regression model (26), with the effect of bias assessed using the fail-safe number method. The fail-safe number was the number of studies that we would need to have missed for our observed result to be nullified to statistical non-significance at the P<0.05 level. Publication bias is generally regarded as a concern if the fail-safe number is less than 5n+10, with n being the number of studies included in the meta-analysis (27). All analyses were performed with Comprehensive Meta-analysis (version 2.0).
Results
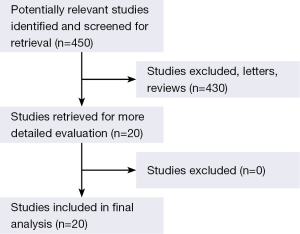
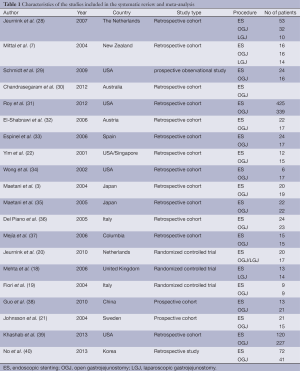
The original search strategy retrieved studies (Figure 1). The abstracts were reviewed and after applying the inclusion and exclusion criteria, articles were selected for full-text evaluation. Of the articles selected, only 20 met full criteria for analysis and are summarised in Table 1. The years of publication ranged from 2001 to 2013.
Full table
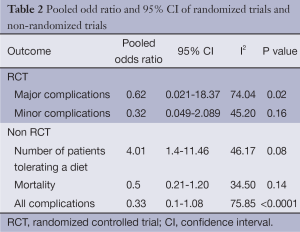
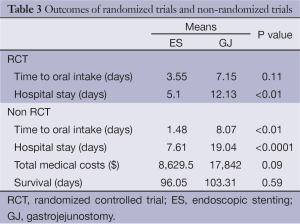
The results of the three randomized controlled trials (RCTs) demonstrated that SEMS resulted in lower major [odds ratio (OR): 0.62, 95% CI: 0.021-18.371] and minor (OR: 0.32, 95% CI: 0.049-2.089) complications in a shorter time to tolerating an oral intake (SEMS: 3.55 days and GJ: 7.15 days) and shorter hospital stay (SEMS: 5.1 days and GJ: 12.13 days). Among the non RCTs: SEMS resulted in a shorter time to tolerating an oral intake (SEMS: 1.48 days and GJ: 8.07 days), lesser complications (OR: 0.33, 95% CI: 0.1-1.08), lower mortality (OR: 0.5, 95% CI: 0.21-1.20) and a shorter hospital stay (SEMS: 7.61 days and GJ: 19.04 days). There was no significant difference between median survival times among RCTs and non RCTs (Tables 2 and 3).
Full table
Full table
Heterogeneity and publication bias
No publication bias was detected using the Egger’s regression model.
Discussion
GOO secondary to unresectable primary or metastatic malignancy is a challenging aspect of patient care. The main objective of a palliative procedure in patients with malignant GOO is to restore their ability to eat.
A comprehensive review of 32 case series including 606 patients was conducted by Dormann et al. (41) 94% of the patients were unable to take food orally or were mainly ingesting liquids. Stent placement and deployment were successful in 589 of the patients (97%). Clinical success was achieved in 526 patients in the group in which technical success was reported (89%; 87% of the entire group undergoing stenting). Disease-related factors accounted for the majority of clinical failures. Oral intake became possible in all of the patients in whom a successful procedure was carried out, with 87% taking soft solids or a full diet, with final resolution of symptoms occurring after a mean of 4 days. There was no procedure-related mortality. Severe complications (bleeding and perforation) were observed in seven patients (1.2%). Stent migration was reported in 31 patients (5%). Stent obstruction occurred in 104 cases (18%), mainly due to tumor infiltration. The mean survival period was 12.1 weeks.
Current literature included three RCTs that compared ES with GJ (18-20). These three trials combined consist of a total of 84 patients. Confounding variables could not be studied in most of the published trials to avoid overfitting. However, if factors, such as chemoradiation therapy, carcinomatosis, age, comorbidities, etc., are not accounted for, results may be biased.
Johns Hopkins, Baltimore (39) recently published a cohort of 347 patients. Technical success was higher for GJ (99% vs. 96%, P=0.004). Complication rates were higher in the GJ group (22.10% vs. 11.66%, P=0.02). Reintervention was more common with ES (adjusted OR 9.18, P<0.0001). Mean LOHS was shorter (adjusted P=0.005) in the ES compared with the GJ group. However, mean hospital charges, including reinterventions, were greater in the ES group (US34,250 vs. US27,599, P=0.03). ES and GJ had comparable reintervention-free time in patients who had reintervention (88 vs. 106 days, respectively, P=0.79). Chemotherapy [adjusted hazard ratio (HR) 3>0.57, P=0.04] and radiation therapy (adjusted HR 0.35, P=0.03) were associated with significantly longer duration of oral intake after ES or GJ.
Boston Scientific Corporation (31) evaluated 425 stenting and 339 GJ hospitalizations. Compared with GJ, median LOS (8 vs. 16 days; P<0.0001) and median cost (US15,366 vs. US27,391; P<0.0001) per claim were both significantly lower for stenting. Stenting was more commonly performed at urban versus rural hospitals (89% vs. 11%; P<0.0001), teaching versus non-teaching hospitals (59% vs. 41%, P=0.0005), and academic institutions (56% vs. 44%; P=0.0157). The institutional patient data analysis included 29 patients who underwent stenting and 75 who underwent surgical GJ. While both modalities were technically successful and relieved GOO in all cases, compared with surgical GJ, the median post-procedure LOS was significantly lower for enteral stenting (1.5 vs. 10.7 days, P<0.0001). There was no difference in rates of delayed complications between stenting and surgical GJ (13.8% vs. 6.7%; P=0.26).
Memorial Sloan-Kettering Cancer Center (29) performed a prospective observational study examining quality of life in patients with malignant GOO. Median overall survival was 64 days. A shorter hospital stay and trend to lower mortality were observed after stent placement; solid food intake and rates of secondary intervention were comparable. Both stent and surgical bypass were associated with acceptable QOL outcomes. Fifteen patients refused participation at 1 month and 28 died of disease before 3 months, so ten patients completed all surveys.
Conclusions
In conclusion, while the technical and clinical outcomes of GJ and stent placement appear comparable in relieving obstruction, stent placement is associated with shorter LOS. This endoscopic approach is also in line with the minimally invasive goals of palliation, namely minimizing pain, hospitalization, and physiologic stress to the patient.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Lopera JE, Brazzini A, Gonzales A, et al. Gastroduodenal stent placement: current status. Radiographics 2004;24:1561-73. [PubMed]
- Warshaw AL, Fernández-del Castillo C. Pancreatic carcinoma. N Engl J Med 1992;326:455-65. [PubMed]
- Maetani I, Tada T, Ukita T, et al. Comparison of duodenal stent placement with surgical gastrojejunostomy for palliation in patients with duodenal obstructions caused by pancreaticobiliary malignancies. Endoscopy 2004;36:73-8. [PubMed]
- Mukherjee S, Kocher HM, Hutchins RR, et al. Palliative surgical bypass for pancreatic and peri-ampullary cancers. J Gastrointest Cancer 2007;38:102-7. [PubMed]
- Lesurtel M, Dehni N, Tiret E, et al. Palliative surgery for unresectable pancreatic and periampullary cancer: a reappraisal. J Gastrointest Surg 2006;10:286-91. [PubMed]
- Van Heek NT, De Castro SM, van Eijck CH, et al. The need for a prophylactic gastrojejunostomy for unresectable periampullary cancer: a prospective randomized multicenter trial with special focus on assessment of quality of life. Ann Surg 2003;238:894-902. [PubMed]
- Mittal A, Windsor J, Woodfield J, et al. Matched study of three methods for palliation of malignant pyloroduodenal obstruction. Br J Surg 2004;91:205-9. [PubMed]
- Jeurnink SM, Polinder S, Steyerberg EW, et al. Cost comparison of gastrojejunostomy versus duodenal stent placement for malignant gastric outlet obstruction. J Gastroenterol 2010;45:537-43. [PubMed]
- Bergamaschi R, Marvik R, Thoresen JE, et al. Open versus laparoscopic gastrojejunostomy for palliation in advanced pancreatic cancer. Surg Laparosc Endosc 1998;8:92-6. [PubMed]
- Denley SM, Moug SJ, Carter CR, et al. The outcome of laparoscopic gastrojejunostomy in malignant gastric outlet obstruction. Int J Gastrointest Cancer 2005;35:165-9. [PubMed]
- Lindsay JO, Andreyev HJ, Vlavianos P, et al. Self-expanding metal stents for the palliation of malignant gastroduodenal obstruction in patients unsuitable for surgical bypass. Aliment Pharmacol Ther 2004;19:901-5. [PubMed]
- van Hooft JE, Uitdehaag MJ, Bruno MJ, et al. Efficacy and safety of the new WallFlex enteral stent in palliative treatment of malignant gastric outlet obstruction (DUOFLEX study): a prospective multicenter study. Gastrointest Endosc 2009;69:1059-66. [PubMed]
- Nassif T, Prat F, Meduri B, et al. Endoscopic palliation of malignant gastric outlet obstruction using self-expandable metallic stents: results of a multicenter study. Endoscopy 2003;35:483-9. [PubMed]
- Maetani I, Tada T, Shimura J, et al. Technical modifications and strategies for stenting gastric outlet strictures using esophageal endoprostheses. Endoscopy 2002;34:402-6. [PubMed]
- Kim TO, Kang DH, Kim GH, et al. Self-expandable metallic stents for palliation of patients with malignant gastric outlet obstruction caused by stomach cancer. World J Gastroenterol 2007;13:916-20. [PubMed]
- Dumas R, Demarquay JF, Caroli-Bosc FX, et al. Palliative endoscopic treatment of malignant duodenal stenosis by metal prosthesis. Gastroenterol Clin Biol 2000;24:714-8. [PubMed]
- Adler DG, Baron TH. Endoscopic palliation of malignant gastric outlet obstruction using self-expanding metal stents: experience in 36 patients. Am J Gastroenterol 2002;97:72-8. [PubMed]
- Mehta S, Hindmarsh A, Cheong E, et al. Prospective randomized trial of laparoscopic gastrojejunostomy versus duodenal stenting for malignant gastric outflow obstruction. Surg Endosc 2006;20:239-42. [PubMed]
- Fiori E, Lamazza A, Volpino P, et al. Palliative management of malignant antro-pyloric strictures. Gastroenterostomy vs. endoscopic stenting. A randomized prospective trial. Anticancer Res 2004;24:269-71. [PubMed]
- Jeurnink SM, Steyerberg EW, van Hooft JE, et al. Surgical gastrojejunostomy or endoscopic stent placement for the palliation of malignant gastric outlet obstruction (SUSTENT study): a multicenter randomized trial. Gastrointest Endosc 2010;71:490-9. [PubMed]
- Johnsson E, Thune A, Liedman B. Palliation of malignant gastroduodenal obstruction with open surgical bypass or endoscopic stenting: clinical outcome and health economic evaluation. World J Surg 2004;28:812-7. [PubMed]
- Yim HB, Jacobson BC, Saltzman JR, et al. Clinical outcome of the use of enteral stents for palliation of patients with malignant upper GI obstruction. Gastrointest Endosc 2001;53:329-32. [PubMed]
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 2009;62:1006-12. [PubMed]
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Controlled Clinical Trials 1986;7:177-88. [PubMed]
- Higgins JP, Thompson SG, Deeks JJ, et al. Measuring inconsistency in meta-analyses. BMJ 2003;327:557-60. [PubMed]
- Egger M, Smith GD, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315:629-34. [PubMed]
- Orwin RG. A fail-safe N for effect size in meta-analysis. J Educ Behav Stat 1983;8:157-9.
- Jeurnink SM, Steyerberg EW, Hof G, et al. Gastrojejunostomy versus stent placement in patients with malignant gastric outlet obstruction: a comparison in 95 patients. J Surg Oncol 2007;96:389-96. [PubMed]
- Schmidt C, Gerdes H, Hawkins W, et al. A prospective observational study examining quality of life in patients with malignant gastric outlet obstruction. Am J Surg 2009;198:92-9. [PubMed]
- Chandrasegaram MD, Eslick GD, Mansfield CO, et al. Endoscopic stenting versus operative gastrojejunostomy for malignant gastric outlet obstruction. Surg Endosc 2012;26:323-9. [PubMed]
- Roy A, Kim M, Christein J, et al. Stenting versus gastrojejunostomy for management of malignant gastric outlet obstruction: comparison of clinical outcomes and costs. Surg Endosc 2012;26:3114-9. [PubMed]
- El-Shabrawi A, Cerwenka H, Bacher H, et al. Treatment of malignant gastric outlet obstruction: endoscopic implantation of self-expanding metal stents versus gastric bypass surgery. European Surgery 2006;38:451-5.
- Espinel J, Sanz O, Vivas S, et al. Malignant gastrointestinal obstruction: endoscopic stenting versus surgical palliation. Surg Endosc 2006;20:1083-7. [PubMed]
- Wong YT, Brams DM, Munson L, et al. Gastric outlet obstruction secondary to pancreatic cancer: surgical vs endoscopic palliation. Surg Endosc 2002;16:310-2. [PubMed]
- Maetani I, Akatsuka S, Ikeda M, et al. Self-expandable metallic stent placement for palliation in gastric outlet obstructions caused by gastric cancer: a comparison with surgical gastrojejunostomy. J Gastroenterol 2005;40:932-7. [PubMed]
- Del Piano M, Ballare M, Montino F, et al. Endoscopy or surgery for malignant GI outlet obstruction? Gastrointest Endosc 2005;61:421-6. [PubMed]
- Mejía A, Ospina J, Muñoz A, et al. Paliación de la obstrucción gastroduodenal maligna. Revista Colombiana de Gastroenterologia 2006;21:19-21.
- Guo JJ, Liang WX, Zhang T. A prospective comparative study of three treatment options in patients with malignant gastric outlet obstruction. Zhonghua Wei Chang Wai Ke Za Zhi 2010;13:598-600. [PubMed]
- Khashab M, Alawad AS, Shin EJ, et al. Enteral stenting versus gastrojejunostomy for palliation of malignant gastric outlet obstruction. Surg Endosc 2013;27:2068-75. [PubMed]
- No JH, Kim SW, Lim CH, et al. Long-term outcome of palliative therapy for gastric outlet obstruction caused by unresectable gastric cancer in patients with good performance status: endoscopic stenting versus surgery. Gastrointest Endosc 2013;78:55-62. [PubMed]
- Dormann A, Meisner S, Verin N, et al. Self-expanding metal stents for gastroduodenal malignancies: systematic review of their clinical effectiveness. Endoscopy 2004;36:543-50. [PubMed]