HDR brachytherapy for anal cancer
Introduction
Anal cancer is a rare disease. Only 1-2% of all gastrointestinal tumors are anal cancer (1). In former times surgery with abdominoperineal resection (APR) followed by colostomy was the first choice of treatment. APR leads to a local control rate after 5 years of 40-75% (2-4). Over the last decades a treatment shift from radical surgery to conservative treatment has occurred. In the beginning of the 1970s, Nigro introduced a novel combined modality treatment (5). In the following years several studies showed the effectiveness of combined radio chemotherapy using EBRT with 5-FU and Mitomycin C (MMC) (5). Conventional 3-D conformal (3DCRT) treatment planning showed a high local control rate. Nowadays intensity modulated radiotherapy (IMRT) should be used for improved treatment results and reduction in toxicity (6-10). But not only EBRT alone is an accepted treatment method, brachytherapy (BT) also has an important role as local dose escalation (boost) in the treatment of anal cancer. Due to the physical and biological advantages BT allows to the physician to apply higher doses direct in the tumor or tumor bed with less toxicity. At the beginning BT target volume definition was based on the clinical findings. Since the introduction of transrectal ultrasound (TRUS) as well of magnetic resonance imaging (MRI), image based implants are possible, resulting in a high precision therapy (9). The different BT methods, limitations and results are discussed in this work.
Principles of BT
At the beginning of the modern BT era most BT implants have been carried out manually. At first catheters, hollow needles or applicators were inserted into the palpable tumor. Later on, BT sources were manually introduced into the previously implanted applicator. Up-to-date remote afterloading machines are now in use. A single miniaturized source connected to a steel wire moves step by step through the applicator, steered by an individual computer program to achieve the calculated volume dose distribution. An advantage as the steep dose fall-off around the source makes it possible to individually increase the tumor dose and to spare the surrounding normal tissue. Short overall treatment time (OTT) also makes BT attractive for patients. The most frequently applied BT methods are high-dose-rate (HDR) BT, low-dose-rate (LDR) or pulsed-dose-rate (PDR) BT. These methods are characterized by the different dose rates delivered by the radiation sources (LDR/PDR: 0.5-1 Gy/h; HDR: >12 Gy/h). The isotope Iridium-192 (Ir192) is commonly used for remote afterloading procedures. Application forms are contact BT, intracavitary-, intraluminal- and endovascular implantations as well interstitial BT. Interstitial BT is an invasive procedure and requires local or general anesthesia. The use of image guided and adapted BT allows a better target volume definition and improves the quality of implants.
Anal cancer BT limitations
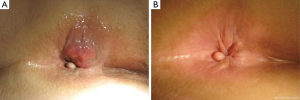
In order to preserve the sphincter function there are some limitations for the use of BT in anal cancer. Not more than the half of the circumference should be implanted. The maximum longitudinal length should be not more than 5 cm. The thickness of the tumor usually should not extend 10 mm (Figure 1A,B) (11).
Methods
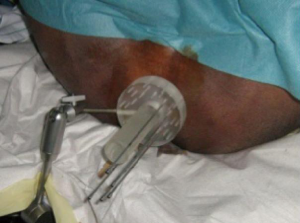
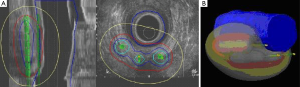
All interstitial implants are performed under local or general anesthesia in lithotomy position. Before the imaging era palpation was the only method to define the target. By digital examination the extent of the tumor was analyzed and hollow steel needles implanted parallel with an interspacing of 1-1.5 cm (Figure 2). Single plane or double plane implants are used depending on the tumor thickness. A ring template is often used for a better guidance of the needle. The needles are placed 1-2 cm beyond the longitudinal extent of the tumor and should cover the whole circumference of the tumor (11). Image based implants become possible by the use of TRUS, computed tomography (CT) or MRI. Of these, implants are most easily done by TRUS. The image guided implantation represents the same procedure that is used for interstitial implant of prostate cancer. Under ultrasound control the needles are implanted directly into the tumor or tumor bed and real time treatment planning is possible. Imaging allows for control of the dose distribution and ensures that the whole tumor is covered by the reference isodose (9,12,13). By using an anal obturator or dilator, the dose to the healthy side of the anal canal be limited (Figure 3A,B).
CT or MRI based implants are possible alternatives, but limited by high technical effort, or by MRI-suitable equipment and lack of real time treatment planning (Figure 3).
Another option is the intraluminal BT. A shielded cylinder is placed in the anal canal under endoscopy view. The treatment planning is then performed based on MRI (14).
Treatment results
LD-BT is well covered in the literature. Papillion et al. published the results of 221 patients (pts) with epidermoid anal cancer. Two months after radio chemotherapy (5-FU and MMC) an interstitial boost using a minimum dose of 15-20 Gy was given in 15-28 hours. The anal preservation rate was 61% and the 5-year survival rate was 65%. In more than 90% of the surviving patients, normal anal function could be preserved (12,14,15). Berger et al. report in a retrospective analysis of 69 pts treated with 40 Gy EBRT followed by 20 Gy interstitial LDR-BT 6 weeks after EBRT the CR was 81% (16). After 5 years the local control rate was 59% and the colostomy rate was 33%. Forty-five pts received a 5-FU-MMC based chemotherapy (13). In the CORS-03 study 162 pts had been analyzed regarding the boost strategy. After a EBRT of 45 Gy 76 pts received an EBRT boost with a mean dose of 18.3 Gy (range, 8-25 Gy) and 86 pts underwent a LDR boost with 17.4 Gy (range, 10-25 Gy). The local recurrence rate after 5 years was 33% for the EBRT arm and 12% for the BT arm (17). In a subgroup analysis of the CORS-03 trial, 99 pts with lymph node metastases (67 pts perirectal, 32 pts ilic and/or inguinal) after 45 Gy EBRT, 49 pts had an EBRT boost with 18.8 Gy (range, 14-25 Gy) and 50 pts had a BT boost with 17.2 Gy (range, 10-25 Gy). Eighteen pts suffered from a local recurrence in the anal canal. The 5-year cumulative rate of local recurrence (CRLR) was 11% for the BT arm and 32% for the EBRT arm. The 5-year overall survival (OS) was 75.5% and 73.3% for the BT group and the EBRT group (18), suggesting that nodal involvement is not a contraindication to BT boost. Another study published in 2007 compared an EBRT boost versus a BT boost, 37 pts. After EBRT of 45 Gy, 37 pts received an EBRT boost of 14.4 Gy in 8 fractions and 47 pts were treated with 14 Gy in 7 fractions within 3 days. The authors didn’t find any difference in cause specific survival (CSS) and OS for all patients after 10 years. The local failure rate at 5 years was 10.3% (BT boost) and 15.4% (EBRT boost). A subgroup analysis showed for localized stage I-II tumors a local control of 100% for the BT arm (19).
The Kiel Group published the results of the first 50 pts treated with a TRUS-guided interstitial HDR-boost in 2005. After an EBRT dose of 45 Gy a dose of 2×4 Gy was applied within 6 weeks after EBRT. The 5-year OS was 74% and the disease specific survival DSS was 82%. Ninety two percent of the pts demonstrated a CR after finishing the treatment. Only three pts had an incomplete or absence tumor response (9). An updated analysis from Kiel described 104 pts with a mean follow up of 10 years. Local control was 89% (93/104) and OS was 93% (96/104). In a subgroup analysis the authors analyzed the pts regarding to pre-planned TRUS—guided implants versus real-time—planning implants. The CSS was 91.5% for the real-time planned group and 86% for the pre-planned group (20).
Toxicity
Only a few data are available comparing the acute and late toxicity of EBRT and BT boost. In a subgroup analysis of a retrospective single-institution study, Oehler-Jaenne et al. analyzed pre-planned TRUS—guided implants versus real-time—planning implants (19). Severe diarrhea was seen in two patients treated with BT boost and in three patients treated with EBRT boost. Chronic proctitis grade >2 was seen in 19% (BT boost) vs. 32% (EBRT boost) and incontinence grades I, II in 18% (BT boost) vs. 28% (EBRT boost). The analysis of Saarilathi et al. compared IMRT verus 3D CRT. Thirteen of twenty-two patients in the IMRT group experienced grade II GI toxicity. In the 3D CRT group, 22/39 had a grade II and 12/39 had a grade III GI toxicity. For the perineal mucosa and skin 4/20 in the IMRT group and 7/39 in the 3D CRT group had a grade I reaction and 16/20 and 32/39 respectively compared to those having received it by HDR BT (n=20). Nine cases of grade II proctitis (23% overall) were observed, seven in the external radiotherapy group and two in the HDR group respectively (21). The long term results from Oblak et al. showed in the majority of the cases (58.2% of patients) grades III, IV radio dermatitis was the predominant acute toxicity. Grades III, IV late toxicity included late anal stenosis in 3.8%, chronic ulceration in 2.5% and incontinence in 8.8%. Those patients treated with a BT boost instead of an EBRT Boost had less toxicity; however, the difference was statistically non-significant (22). Doniec et al. reported three pts (6%) with a late incontinence as the only late radiotherapy related toxicity. Two of them underwent colostomy (12).
Time schedule of combined EBRT + BT boost
The OTT and the time gap between EBRT and BT boost are prognostic factors for the local control rate. If the time gap between EBRT and Boost is >37.5 days the local control is less than compared to the pts with a <37.5 days gap (22,23).In the CORS-03 Hannoun-Levy showed the influence of OTT. If the OTT is >80 days the local control rate is not influenced by the boost technique. But if the OTT is shortened to <80 days local control is increased significantly using the BT boost instead an EBRT boost (17).
Conclusions
Definitive combined radio chemotherapy is the current standard for function preservation treatment of anal cancer. IMRT techniques should be used instead of 3D treatment. If the tumor is eligible for BT, image guidance is recommended in BT target definition and to guide the implantation procedure. In the hand of experienced personnel a HDR BT boost is safe, maximally individualized and represents an effective method with high quality assurance (QA) potential of the procedure. Limitations for BT are large tumors with an extension of more than 5 cm and/or more of the half of the circumference. In the majority of cases anal cancer BT is performed 2-3 weeks after EBRT using 2 fractions of 4-6 Gy each. Time constraints needs to be followed to reach the maximal potential of this elegant type of treatment delivery.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Tumorregister München. Available online: http://www.tumorregister-muenchen.de/facts/base/base_C21__G.pdf
- Boman BM, Moertel CG, O’Connell MJ, et al. Carcinoma of the anal canal. A clinical and pathologic study of 188 cases. Cancer 1984;54:114-25. [PubMed]
- Dougherty BG, Evans HL. Carcinoma of the anal canal: a study of 79 cases. Am J Clin Pathol 1985;83:159-64. [PubMed]
- Greenall MJ, Quan SH, Urmacher C, et al. Treatment of epidermoid carcinoma of the anal canal. Surg Gynecol Obstet 1985;161:509-17. [PubMed]
- Nigro ND, Vaitkevicius VK, Considine B Jr. Combined therapy for cancer of the anal canal: a preliminary report. Dis Colon Rectum 1974;17:354-6. [PubMed]
- Kachnic LA, Winter K, Myerson RJ, et al. RTOG 0529: a phase 2 evaluation of dose-painted intensity modulated radiation therapy in combination with 5-fluorouracil and mitomycin-C for the reduction of acute morbidity in carcinoma of the anal canal. Int J Radiat Oncol Biol Phys 2013;86:27-33. [PubMed]
- Milano MT, Jani AB, Farrey KJ, et al. Intensity-modulated radiation therapy (IMRT) in the treatment of anal cancer: toxicity and clinical outcome. Int J Radiat Oncol Biol Phys 2005;63:354-61. [PubMed]
- Menkarios C, Azria D, Laliberté B, et al. Optimal organ-sparing intensity-modulated radiation therapy (IMRT) regimen for the treatment of locally advanced anal canal carcinoma: a comparison of conventional and IMRT plans. Radiat Oncol 2007;2:41. [PubMed]
- Löhnert M, Doniec JM, Kovács G, et al. New method of radiotherapy for anal cancer with three-dimensional tumor reconstruction based on endoanal ultrasound and ultrasound-guided afterloading therapy. Dis Colon Rectum 1998;41:169-76. [PubMed]
- Khosla D, Kumar R, Kapoor R, et al. Sphincter preservation in anal cancer: a brief review. Saudi J Gastroenterol 2013;19:101-7. [PubMed]
- Mazeron JJ, Van Limbergen E. Anorectal Cancer. In: Gerbaulet A, Pötter R, Mazeron JJ, et al. eds. The GEC ESTRO Handbook of Brachytherapy. Brussels: ESTRO, 2002:505-14.
- Doniec JM, Schniewind B, Kovács G, et al. Multimodal therapy of anal cancer added by new endosonographic-guided brachytherapy. Surg Endosc 2006;20:673-8. [PubMed]
- Papillon J, Montbarbon JF. Epidermoid carcinoma of the anal canal. A series of 276 cases. Dis Colon Rectum 1987;30:324-33. [PubMed]
- Vordermark D, Flentje M, Sailer M, et al. Intracavitary afterloading boost in anal canal carcinoma. Results, function and quality of life. Strahlenther Onkol 2001;177:252-8. [PubMed]
- Papillon J, Montbarbon JF, Gerard JP, et al. Interstitial curietherapy in the conservative treatment of anal and rectal cancers. Int J Radiat Oncol Biol Phys 1989;17:1161-9. [PubMed]
- Berger C, Félix-Faure C, Chauvet B, et al. Conservative treatment of anal canal carcinoma with external radiotherapy and interstitial brachytherapy, with or without chemotherapy: long-term results. Cancer Radiother 1999;3:461-7. [PubMed]
- Hannoun-Levi JM, Ortholan C, Resbeut M, et al. High-dose split-course radiation therapy for anal cancer: outcome analysis regarding the boost strategy (CORS-03 study). Int J Radiat Oncol Biol Phys 2011;80:712-20. [PubMed]
- Moureau-Zabotto L, Ortholan C, Hannoun-Levi JM, et al. Role of brachytherapy in the boost management of anal carcinoma with node involvement (CORS-03 study). Int J Radiat Oncol Biol Phys 2013;85:e135-42. [PubMed]
- Oehler-Jänne C, Seifert B, Lütolf UM, et al. Clinical outcome after treatment with a brachytherapy boost versus external beam boost for anal carcinoma. Brachytherapy 2007;6:218-26. [PubMed]
- Niehoff P, Schumacher N, Siebert FA, et al. TRUS guided interstitial HDR Brachytherapy combined with RCT for treatment of anal cancer. Radiother Oncol 2014;111:420.
- Saarilahti K, Arponen P, Vaalavirta L, et al. The effect of intensity-modulated radiotherapy and high dose rate brachytherapy on acute and late radiotherapy-related adverse events following chemoradiotherapy of anal cancer. Radiother Oncol 2008;87:383-90. [PubMed]
- Oblak I, Petric P, Anderluh F, et al. Long term outcome after combined modality treatment for anal cancer. Radiol Oncol 2012;46:145-52. [PubMed]
- Weber DC, Kurtz JM, Allal AS. The impact of gap duration on local control in anal canal carcinoma treated by split-course radiotherapy and concomitant chemotherapy. Int J Radiat Oncol Biol Phys 2001;50:675-80. [PubMed]