
Intracorporeal anastomosis versus extracorporeal anastomosis for minimally invasive colectomy
Introduction
Laparoscopic techniques were adopted by general surgeons in the 1980s with subsequent adaptation to colorectal surgery in 1991 (1). The robotic approach was introduced to colorectal surgery in 2002 (2,3) and upgrades in the robotic platform have resulted in recent increased adoption by colorectal surgeons. Subsequent studies demonstrated improved minimally invasive short-term post-operative outcomes, with earlier return of bowel function, lower analgesia requirements, and shorter hospital length of stay (4,5). Data have also demonstrated equivalent oncologic outcomes for patients undergoing minimally invasive colon resections when compared to open operations, with associated decreased post-operative morbidity (6,7). Recent data suggest a continued increase in the use of minimally invasive approaches for both benign and malignant colorectal disease (8).
How to restore intestinal continuity and create a superior anastomosis is still a topic for consideration in colorectal surgery. Minimizing infectious complications that include anastomotic leak, post-operative abscess, and surgical site infection is imperative, but decreasing time to discharge and quicker recovery are also key outcomes. Minimally invasive ileocolic and colorectal anastomoses may be performed using intracorporeal (or total laparoscopic/robotic) or extracorporeal (or laparoscopic/robot assisted) techniques. This article is a review of operative techniques for both intracorporeal anastomosis (IA) and extracorporeal anastomosis (EA), and an examination of published data comparing each approach.
Descriptions and critiques of anastomotic techniques
Extracorporeal anastomosis
Anastomotic technique
For right colectomies, after complete mobilization of the terminal ileum, cecum, ascending colon, and proximal transverse colon, an upper midline incision is typically made by extending the camera port incision along the midline. The mobilized ileum and colon are then exteriorized after insertion of a wound protector. The terminal ileum and transverse colon are divided and the ileocolic anastomosis is constructed using standard open techniques. Some surgeons divide the mesentery and vessels laparoscopically or robotically prior to exteriorizing the specimen and performing the extracorporeal resection and anastomosis. Others perform various parts of mesenteric and vessel division after exteriorization of the specimen and prior to bowel division and anastomosis.
After division of the specimen, an extracorporeal functional end-to-end anastomosis is then constructed by aligning the ileum and transverse colon in an isoperistaltic or antiperistaltic fashion. Enterotomies are made in the ileum and transverse colon and a linear cutter 55 or 75 mm blue load stapler is placed through these enterotomies and fired, thereby creating the anastomosis. The common enterotomy is then sutured closed in one or two layers.
For left colectomies, after adequate mobilization, the upper rectum is transected using a laparoscopic or robotic linear stapler. A midline or Pfannenstiel incision is made, a wound protector is placed, and the proximal end of the transected bowel is delivered through the incision thereby exteriorizing the specimen still attached to the proximal colon. The specimen is resected using standard open techniques. A circular stapler anvil is secured in the open proximal end of soft and pliable colon using a purse string suture. The colon with secured anvil is returned to the abdominal cavity and pneumoperitoneum reinstituted. The circular stapler is passed through the anus and coupled with the anvil in the proximal colon, and an end-to-end anastomosis is created either laparoscopically or with robotic assistance. The anastomosis is tested for leaks using endoscopic air insufflation with saline in the pelvis, and endoscopic viability is confirmed on both sides of the anastomosis.
Extracorporeal anastomosis advantages
Exteriorization of the proximal and distal bowel with attached specimen allows for visual inspection and palpation prior to resection and anastomosis. Some surgeons think this may help confirm healthy, soft and viable bowel for anastomosis and confirm appropriate margins for oncologic resections. Since bowel transection and creation of the anastomosis occur outside of the abdominal cavity, there is potentially a decrease in risk of intra-abdominal spillage of colonic contents related to this technique.
Extracorporeal anastomosis disadvantages
Because of the need to exteriorize the bowel for resection and anastomosis, significantly more bowel and mesentery must be mobilized to obtain adequate reach for specimen resection and anastomosis, especially in obese patients with thick abdominal walls. For right hemicolectomies, exteriorizing the transverse colon to a small midline specimen extraction site may be challenging with risk of traction injury to bowel or mesentery. This may result in serosal injuries, mesenteric bleeding, and devascularization of bowel and mesentery that may potentially contribute to post-operative ileus (9). The transverse colon may not reach the extraction site easily and the midline incision may need to be lengthened to accommodate. Many surgeons use a midline extraction site for extracorporeal right colectomies. The midline site is associated with a significantly higher rate of incisional hernias (8–12%) with associated long-term morbidity when compared to off-midline extraction sites (10,11).
Intracorporeal anastomosis
Anastomotic technique
For right colectomies, an intracorporeal ileo-transverse colon anastomosis is typically chosen for the minimally invasive approach (see Figure 1). Following complete medial-to-lateral and lateral-to medial mobilization of the ileum and colon, and complete detachment of the mesentery from the retroperitoneum, the terminal ileum and transverse colon are transected using a laparoscopic or robotic linear cutter stapler. The ileum and transverse colon are aligned in either an isoperistaltic or antiperistaltic configuration. A seromuscular stay suture is placed between the ileum and transverse colon and retracted toward the right side of the abdomen to optimize alignment. A colotomy and enterotomy are then made and the linear cutter stapler is placed and fired, creating the anastomosis. The common enterotomy is then closed with suture in one or two layers. The specimen is then removed via a small Pfannenstiel or other off-midline incision.
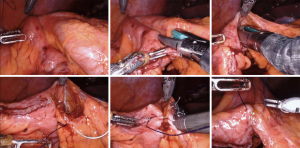
When describing anastomotic options for left colectomies, it may be best to refer to intracorporeal technique rather than intracorporeal anastomosis (12). The anastomosis is “intracorporeal” for both intracorporeal and extracorporeal techniques. However, the entire operation prior to specimen extraction that includes mobilization of the mesentery and division of the vessels, upper rectum, and proximal colon, followed by placement of the anvil and creation of the anastomosis, are all done within the abdomen for the intracorporeal technique (see Figure 2). In contrast, the extracorporeal left colectomy technique is characterized by resection of the specimen and placement of the anvil after delivering the specimen through the extraction site incision using standard open techniques.
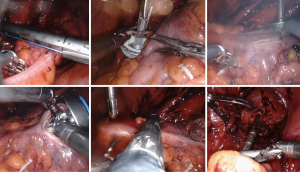
The intracorporeal technique starts with medial-to-lateral mobilization of the descending and sigmoid colon and mesentery. The inferior mesenteric vessels are divided after identification of the left ureter. Lateral-to-medial mobilization of the left colon is followed by intracorporeal division of the upper rectum using a laparoscopic or robotic linear cutter stapler. The mesentery is divided from point of transection of the inferior mesenteric artery to the proposed point of transection of the descending colon. A long 3–4 cm colotomy is made distal, and a small 6 mm colotomy made proximal to the proposed point of transection of the descending colon. The anvil is introduced into the abdomen through either a Pfannenstiel or other off-midline extraction site incision. After re-establishing pneumoperitoneum, the anvil is passed through the long colotomy and then the shaft of the anvil routed through the small colotomy proximal to the proposed point of transection. The descending colon is then divided with a linear cutter stapler after confirming viability with immunofluorescence. The long colotomy, now on the specimen side, is closed with a running suture and the specimen is set aside until completion of the anastomosis. The circular stapler is passed through the anus and coupled with the anvil on the descending colon and a side-of-colon to end-of-rectum anastomosis is created. The specimen is then removed through a Pfannenstiel or other off-midline extraction site incision.
Intracorporeal anastomosis advantages
Since the colon does not require exteriorization for resection and anastomosis, there is no need to mobilize bowel that will remain in-situ, and no traction is required to deliver the specimen to a small extraction site incision. This decreases the risk for mesenteric bleeding, serosal injuries, and the occasional need to extend the extraction site incision. This may result in less ileus (9). The length of the IA extraction site incision is not influenced by whether the colon will reach, and the size of the IA extraction site incision is limited only by the size of the specimen (12). For some morbidly obese patients, the only minimally invasive option may be an IA approach because of the degree of difficulty related to thick and short mesentery reaching an extracorporeal extraction site in a thick abdominal wall (9,12). Because the specimen may be removed at any off-midline location, the risk for incisional hernia and subsequent related long-term morbidity from complex hernia mesh repair is reduced (10,11).
Intracorporeal anastomosis disadvantages
Since the specimen transection is intracorporeal, palpation of the bowel prior to transection is not performed and some consider this a disadvantage. Additionally, the enterotomies for the anastomosis are created intra-abdominally, exposing the peritoneum to potential intraluminal contamination. However, no studies have demonstrated an increase in surgical site infections and sepsis when comparing IA and EA groups (9,12).
Review of published data
Right colectomies
Table 1 displays single and multi-institution studies that compare outcomes for minimally invasive IA and EA right colectomies. Many studies show favorable outcomes for IA, demonstrating decreased conversion-to-open operations, shorter gastrointestinal recovery time, shorter hospital length of stay, and decreased postoperative complications, including both short-term (surgical site infection, anastomotic leak, and ileus) and long term (incisional hernia and small bowel obstruction) complications (9,13,15-18) when compared to EA. Operative times in the IA group were longer than in the EA group in most of these studies (9,15,17).
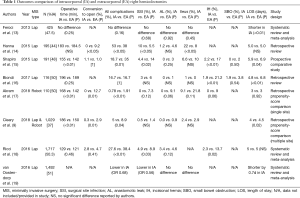
Full table
A propensity-matched comparison of 379 IA right colectomies (335 robotic-assisted and 44 laparoscopic) and 650 EA right colectomies (253 robotic-assisted and 397 laparoscopic) showed significantly lower conversion rates (0.3% vs. 2.9%, P=0.01), shorter hospital length of stay (4.0 vs. 4.5 days, P=0.02) and fewer 30-day postoperative complications (P=0.04) than the EA group (9). Subsequent systematic reviews and meta-analyses have confirmed improved outcomes for laparoscopic IA, with decreased conversion-to-open operations, decreased short and long term post-operative complications, and shorter length of stay when compared to EA (18,19).
Left colectomies
Literature describing the intracorporeal technique for left sided colorectal resections has recently emerged and is the standard technique taught at the national colon and rectal surgery residency robotics course sponsored by the Association of Program Directors for Colon and Rectal Surgery (12,20). Table 2 displays studies that compare IA and EA for minimally invasive left colectomies. These retrospective studies demonstrated faster recovery of bowel function, decreased post-operative complications, and shorter length of stay in the IA group vs. the EA group for patients who underwent minimally invasive left colectomies (21-24). Further, a propensity score-matched comparison of 57 intracorporeal and 57 extracorporeal sigmoid resections showed that there were fewer conversions in the intracorporeal group (5.26% vs. 19.3%, P=0.029). In addition, there were fewer extraction site hernias in the intracorporeal group (0% vs. 10.53%, P=0.027), likely because there were fewer midline extraction site incisions (8.77% vs. 38.6%, P<0.001) in the IA group (12). The authors suggest that the greater number of midline extraction sites in the EA group may be related to the difficulty mobilizing colon with attached specimen to a Pfannenstiel or other off-midline extraction site incision. In addition, extraction sites for conversion-to-open cases are typically midline and there was a higher conversion rate in the EA group (12).
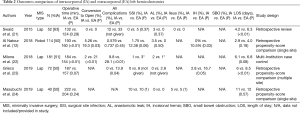
Full table
The technical difficulties encountered with traction injury and mesenteric bleeding while attempting to make the transverse colon reach the extraction site during an extracorporeal right colectomy may also be relevant for extracorporeal sigmoid resections when struggling to mobilize the left colon to an extracorporeal location at the extraction site incision. In contrast to the right colectomy studies comparing IA vs. EA, however, there were no consistent significant differences in time to gastrointestinal recovery, hospital length of stay, 30-day complications, and readmissions between groups in the studies comparing these anastomotic techniques in left colectomies (12,21-24). Additionally, operative time was significantly longer in the IA group (12,22). These studies suggest that the IA approach for sigmoid resection is safe, has some outcomes advantages, and warrants further analysis.
Meta-analysis of right and left colectomies
Emile et al. published a systemic review and meta-analysis that included twenty studies (18 right colectomy and 2 left colectomy) comparing IA versus EA for minimally invasive colectomies. Their analysis included 3,745 patients and confirmed similarly favorable results for the IA group including shorter hospital length of stay, decreased short and long term complication rates, fewer anastomotic leaks, surgical site infections, and incisional hernias (25). Interestingly, the operative time difference between IA and EA groups in this analysis only varied by 13 minutes, suggesting that operative times for the IA approach may improve with experience (25).
There is a paucity of literature comparing robotic and laparoscopic minimally invasive approaches for IA and EA. Studies comparing robotic IA with laparoscopic EA showed favorable outcomes for the robotic approach but it may have been the IA technique rather than the robotic approach responsible for the favorable outcomes (26,27). IA advantages are demonstrated by surgeons capable of minimally invasive suturing techniques. IA with the robotic platform is likely available to more surgeon skill sets than laparoscopic IA because of robotic articulating instruments and ergonomic advantages (9).
Summary
The intracorporeal anastomosis for right and left hemicolectomies has several outcomes advantages when compared to the extracorporeal technique. These intracorporeal technique advantages include the ability to perform the resection and anastomosis without traction injury to bowel remaining in-situ or the need to lengthen the extraction site incision delivering the specimen under tension, and the ability to use an off-midline extraction site incision with less risk for incisional hernias. Further advancements in minimally invasive technology may allow further refinements in intracorporeal techniques that may convey other outcomes advantages.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Nan Zun Teo, James Chi-Yong Ngu) for the series “Current Strategies in Colon Cancer Management” published in Journal of Gastrointestinal Oncology. The article was sent for external peer review organized by the Guest Editors and the editorial office.
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jgo.2019.12.02). The series “Current Strategies in Colon Cancer Management” was commissioned by the editorial office without any funding or sponsorship. Robert K. Cleary has received honoraria from Intuitive Surgical, Inc. for educational speaking. Rebecca F. Brown has no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Fowler DL, White SA. Laparoscopy-assisted sigmoid resection. Surg Laparosc Endosc 1991;1:183-8. [PubMed]
- Weber PA, Merola S, Wasielewski A, et al. Telerobotic-assisted laparoscopic right and sigmoid colectomies for benign disease. Dis Colon Rectum 2002;45:1689-94; discussion 1695-6.
- Hashizume M, Shimada M, Tomikawa M, et al. Early experiences of endoscopic procedures in general surgery assisted by a computer-enhanced surgical system. Surg Endosc 2002;16:1187-91. [Crossref] [PubMed]
- Tjandra JJ, Chan MK. Systematic review on the short-term outcome of laparoscopic resection for colon and rectosigmoid cancer. Colorectal Dis 2006;8:375-88. [Crossref] [PubMed]
- Veldkamp R, Kuhry E, Hop WC, et al. Laparoscopic surgery versus open surgery for colon cancer: short-term outcomes of a randomised trial. Lancet Oncol 2005;6:477-84. [Crossref] [PubMed]
- Colon Cancer Laparoscopic or Open Resection Study Group, Buunen M, Veldkamp R, et al. Survival after laparoscopic surgery versus open surgery for colon cancer: long-term outcome of a randomised clinical trial. Lancet Oncol 2009;10:44-52. [Crossref] [PubMed]
- Kuhry E, Schwenk W, Gaupset R, et al. Long-term outcome of laparoscopic surgery for colorectal cancer: a cochrane systematic review of randomised controlled trials. Cancer Treat Rev 2008;34:498-504. [Crossref] [PubMed]
- Yeo H, Niland J, Milne D, et al. Incidence of minimally invasive colorectal cancer surgery at National Comprehensive Cancer Network centers. J Natl Cancer Inst 2014;107:362. [Crossref] [PubMed]
- Cleary RK, Kassir A, Johnson CS, et al. Intracorporeal versus extracorporeal anastomosis for minimally invasive right colectomy: A multi-center propensity score-matched comparison of outcomes. PLoS One 2018;13:e0206277. [Crossref] [PubMed]
- Harr JN, Juo YY, Luka S, et al. Incisional and port-site hernias following robotic colorectal surgery. Surg Endosc 2016;30:3505-10. [Crossref] [PubMed]
- Samia H, Lawrence J, Nobel T, et al. Extraction site location and incisional hernias after laparoscopic colorectal surgery: should we be avoiding the midline? Am J Surg 2013;205:264-7; discussion 268. [Crossref] [PubMed]
- Al Natour RH, Obias V, Albright J, et al. A propensity score matched comparison of intracorporeal and extracorporeal techniques for robotic-assisted sigmoidectomy in an enhanced recovery pathway. J Robot Surg 2019;13:649-56. [Crossref] [PubMed]
- Feroci F, Lenzi E, Garzi A, et al. Intracorporeal versus extracorporeal anastomosis after laparoscopic right hemicolectomy for cancer: a systematic review and meta-analysis. Int J Colorectal Dis 2013;28:1177-86. [Crossref] [PubMed]
- Hanna MH, Hwang GS, Phelan MJ, et al. Laparoscopic right hemicolectomy: short- and long-term outcomes of intracorporeal versus extracorporeal anastomosis. Surg Endosc 2016;30:3933-42. [Crossref] [PubMed]
- Shapiro R, Keler U, Segev L, et al. Laparoscopic right hemicolectomy with intracorporeal anastomosis: short- and long-term benefits in comparison with extracorporeal anastomosis. Surg Endosc 2016;30:3823-9. [Crossref] [PubMed]
- Biondi A, Santocchi P, Pennestri F, et al. Totally laparoscopic right colectomy versus laparoscopically assisted right colectomy: a propensity score analysis. Surg Endosc 2017;31:5275-82. [Crossref] [PubMed]
- Akram WM, Al-Natour RH, Albright J, et al. A propensity score-matched comparison of intracorporeal and extracorporeal techniques for robotic-assisted right colectomy in an Enhanced Recovery Pathway. Am J Surg 2018;216:1095-100. [Crossref] [PubMed]
- Ricci C, Casadei R, Alagna V, et al. A critical and comprehensive systematic review and meta-analysis of studies comparing intracorporeal and extracorporeal anastomosis in laparoscopic right hemicolectomy. Langenbecks Arch Surg 2017;402:417-27. [Crossref] [PubMed]
- van Oostendorp S, Elfrink A, Borstlap W, et al. Intracorporeal versus extracorporeal anastomosis in right hemicolectomy: a systematic review and meta-analysis. Surg Endosc 2017;31:64-77. [Crossref] [PubMed]
- Disbrow DE, Pannell SM, Shanker BA, et al. The Effect of Formal Robotic Residency Training on the Adoption of Minimally Invasive Surgery by Young Colorectal Surgeons. J Surg Educ 2018;75:767-78. [Crossref] [PubMed]
- Swaid F, Sroka G, Madi H, et al. Totally laparoscopic versus laparoscopic-assisted left colectomy for cancer: a retrospective review. Surg Endosc 2016;30:2481-8. [Crossref] [PubMed]
- Milone M, Angelini P, Berardi G, et al. Intracorporeal versus extracorporeal anastomosis after laparoscopic left colectomy for splenic flexure cancer: results from a multi-institutional audit on 181 consecutive patients. Surg Endosc 2018;32:3467-73. [Crossref] [PubMed]
- Grieco M, Cassini D, Spoletini D, et al. Intracorporeal Versus Extracorporeal Anastomosis for Laparoscopic Resection of the Splenic Flexure Colon Cancer: A Multicenter Propensity Score Analysis. Surg Laparosc Endosc Percutan Tech 2019;29:483-8. [Crossref] [PubMed]
- Masubuchi S, Okuda J, Hamamoto H, et al. Intracorporeal Versus Extracorporeal Anastomosis in Laparoscopic Left Colectomy for Left Sided Colon Cancer: A Retrospective Study. Clin Surg 2019;4:2506.
- Emile SH, Elfeki H, Shalaby M, et al. Intracorporeal versus extracorporeal anastomosis in minimally invasive right colectomy: an updated systematic review and meta-analysis. Tech Coloproctol 2019;23:1023-35. [Crossref] [PubMed]
- Morpurgo E, Contardo T, Molaro R, et al. Robotic-assisted intracorporeal anastomosis versus extracorporeal anastomosis in laparoscopic right hemicolectomy for cancer: a case control study. J Laparoendosc Adv Surg Tech A 2013;23:414-7. [Crossref] [PubMed]
- Trastulli S, Coratti A, Guarino S, et al. Robotic right colectomy with intracorporeal anastomosis compared with laparoscopic right colectomy with extracorporeal and intracorporeal anastomosis: a retrospective multicentre study. Surg Endosc 2015;29:1512-21. [Crossref] [PubMed]


