A genetic database can be utilized to identify potential biomarkers for biphenotypic hepatocellular carcinoma-cholangiocarcinoma
Introduction
Biphenotypic hepatocellular carcinoma-cholangiocarcinoma (HCC-CC) comprises an estimated 1–6.5% of primary liver neoplasms (1). Although this entity does demonstrate pathologic features of its well-established biphenotypic counterparts, the characteristics of this neoplasm make diagnosis challenging using conventional radiologic imaging and serologic markers. Moreover, accurate prognostic information is affected by the low frequency of this tumor, which restricts available clinical information even within large medical centers (2-20).
There are numerous genetic and risk factors important for hepatocellular carcinoma (HCC) and cholangiocarcinoma (CC) separately, which have yet to be addressed in HCC-CC (17-22). Prior studies have examined the role of cytoskeletal stability, apoptosis, and the inflammatory cascade in HCC-CC (4-16). Additionally, as HCC-CC poses a diagnostic dilemma for radiologists, tumor specific biomarker may assist in the diagnosis of this neoplasm and prognostic assessment (23,24).
In this study, we evaluate the utility of a genetic database to identify potential biomarkers for biphenotypic HCC-CC using shared genetic characteristics.
Methods
Variables
We initially evaluated the pathologic subtypes of HCC-CC, HCC and CC (25,26). Pathologic subtypes for HCC-CC included classical and stem-cell. Pathologic subtypes for HCC included: fibrolamellar, scirrhous, sarcomatoid and lympho-epithelial. Subtypes for CC included: intraductal papillary, intestinal-type, clear, squamous and small cell.
After performing our literature search, we identified risk factors relevant to HCC-CC, HCC alone and CC alone (12-15,17-22). Among these risk factors, cirrhosis, hepatitis B virus (HBV) and hepatitis C virus (HCV) viral infections are evaluated risk factors for HCC-CC (12-15,17-20). For HCC alone, the aforementioned risk factors were included as were the following: portal hypertension, alcoholic fatty liver, aflatoxin, peliosis hepatitis, autoimmune hepatitis (AIH), primary biliary cirrhosis (PBC), granulomatous hepatitis, non-alcoholic fatty liver disease (NAFLD), non-alcoholic steatohepatitis (NASH), hemochromatosis (HCM), glycogen storage disease, Wilson’s disease, porphyria cutanea tarda (PCT), alpha-1 antitrypsin, tyrosinemia, portal vein thrombosis, Budd-Chiari syndrome. For CC we evaluated the following variables: primary sclerosing cholangitis (PSC), cystic disease of the liver, biliary cyst, choledochal cyst, Caroli disease, other congenital malformation of the bile ducts, other congenital malformations of the gall bladder, cholangitis, schistosomiasis, opisthorchiasis, clonochiasis, recurrent cholangitis, and biliary stricture.
Genetic database
To evaluate the genetic associations with each variable we utilized GeneCards®, The Human Gene Compendium (http://www.genecards.org) (27-33). Our initial search was performed on HCC alone, CC alone, and finally HCC-CC. Next, we evaluated the risk factors mentioned above utilizing the same compendium database. We then evaluated each possibly biomarkers in accordance to HCC alone, CC alone, and finally HCC-CC. All results were recorded and each gene was scrutinized manually, independent of the search results.
During our manual assessment of each gene we evaluated gene function, pathway and interaction, association with other genes, cellular location, genomic location, and existing therapeutic targets. The results of our search were then entered into the Pathway Interaction Database from the National Cancer Institute (PID-NCI) (http://pid.nci.nih.gov), to generate a biomolecule interaction map.
Results
Overall genetic characteristics
The results of our query yielded 690 genes for HCC, 98 genes for CC and 50 genes for HCC-CC. A summary of the search results for these searches can be visualized in Table S1. Genes depicted in this analysis demonstrate the role of hormonal regulation, embryonic development, cell surface adhesion, cytokeratin stability, mucin production, metalloproteinase regulation, Ras signaling, metabolism and apoptosis. Table 1 depicts the relationship between these genes, the genomic and cellular location. These genes were integrated into a PID-NCI biomolecule interaction map (Figure S1), demonstrating an overview of the interactions between each gene for HCC-CC.
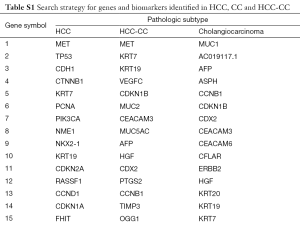
Full table
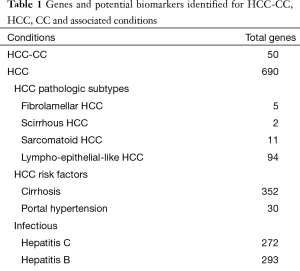
Full table

Gamma-glutamyl transpeptidase (GGT) appeared to have the highest number of associated risk factors (20), followed by the cytokeratin-related genes (KRT7, 8, 9, 18 and 19) (4, 5, 8, 7 respectively). Alkaline phosphatase (ALP) related genes (ALPL, ALPP, ALPPL2) also had a high number of associated etiologies (4, 7 and 4 respectively) (Table 1). The genetic location for genes involved in hormonal regulation demonstrated a genetic location of 11p15. Cell adhesion molecules hepatocyte growth factor (HGF) and mesenchymal epithelial transition (MET) demonstrated a genetic location of 7p and genes involved in mucin production 11p15.5. Overall cytokeratin genes demonstrated a genetic location of 12q13, with the exception of KRT19, with a location of 17q21.
Cell adhesion molecules (HGF and MET) are located in the extracellular matrix (ECM) and cell membrane. Cytokeratin molecules are expressed in the cellular membrane, Golgi apparatus, and nucleus, except for KRT19, located in the ECM and cellular membrane. Mucin production genes MUC2 and MUC5AC localize to the ECM. Metalloproteinase, membrane metallo-endopeptidase (MME) and reversion-inducing-cysteine-rich protein with kazal motifs (RECK), were located in the ECM and cell membrane, while MMP7 and tissue inhibitor of metalloproteinases 3 (TIMP3) were located in the ECM and Nucleus. Gene ALPL, ALPP, ALPPL2 and GGT were located in all cellular locations.
The relationship of these genes to each other is summarized in Table 1 and Figure S1. There appears to be a linkage between the embryonic genes and secretin (SCT). KRT18 also demonstrated some associations with the cell adhesion genes. Not surprising, Ras-signaling genes have a relation with apoptosis genes which in turn overlap with cell adhesion, cytokeratin, metabolism and metalloproteinase genes.
Pathologic subtypes
A complete listing of all genes for each pathologic subtype is presented in Table S2. For each pathologic subtype of HCC-CC, no novel genes could be identified, likely due to extreme search specificity. A detailed examination of HCC subtypes was performed. Fibrolamellar HCC demonstrated no genetic overlap with HCC-CC. Scirrhous HCC possessed two novel genes including MET which shared a common bridge with HCC-CC. Sarcomatoid HCC demonstrated carcinoembryonic antigen-related cell adhesion molecule 3 (CEACAM3) and CHD1 in common with HCC-CC. Among the pathologic subtypes of HCC, lympho-epithelial HCC had the most genetic overlap with HCC-CC possessing both cadherin (CDH1) and MET, along with ALPP, ALPL, ALPPL2, MUC2, MUC5AC, NAT2, PTGS2, SCT, catenin (cadherin-associated protein), alpha 1 (CTNNA1) and vascular endothelial growth factor C (VEGFC). This totaled 12 genes or 24% overlap with HCC-CC.

Full table
Next, the pathologic subtypes of CC were evaluated for overlap with HCC-CC. Intraductal papillary CC shares SMAD family member 4 (SMAD4), Intestinal-type CC possessed Kirsten rat sarcoma viral oncogene homolog (KRAS), KRT7, phosphatidylinositol-4,5-bisphosphate 3-kinase, catalytic subunit alpha (PIK3CA) and tumor protein p53 (TP53) (8% overlap) in common with HCC-CC. Numerically, signet ring CC was similar to the intraductal papillary subtype, differentially expressing 4 genes (8%) in common with HCC-CC (KRT7, MUC2, CDH1 and CEACAM5). Clear cell CC, small cell CC, and squamous cell CC share 20%, 54%, and 78% gene overlap with biphenotypic HCC-CC, respectively. Novel genes expressed by both clear cell CC and HCC-CC include 8-oxoguanine DNA glycosylase (OGG1), CDH1, fragile histidine triad (FHIT) and SMAD family member 4 (SMAD4) among others for 20% overlap with HCC-CC. Genes shared by both HCC-CC and squamous cell CC involve functions such as cytokeratin stability, apoptosis, Ras-signaling, cell adhesion, embryonic development, metalloproteinase, metabolism and tumor necrosis factor alpha pathways.
Hepatocellular carcinoma (HCC) risk factors
Known risk factors for HCC were evaluated with respect to HCC-CC. Table 2 depicts the total number of genes available for each risk factor for HCC when evaluated separately. Cirrhosis had the highest number of genes at 352, followed by 293 for HBV and 272 for HCV. Peliosis hepatitis had the least number of associated genes.
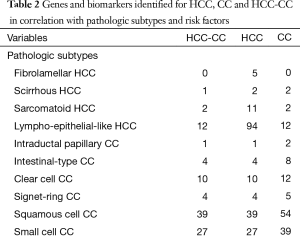
Full table
We compared the genes in each risk factor with HCC and HCC-CC. The results of this comparison are listed in Table 3. Of all the listed risk factors, cirrhosis had the highest number of gene associations with HCC [156] and HCC-CC (19 of 50 or 38%). These genes are summarized in Table S3. Most commonly, cirrhosis and HCC-CC differentially expressed genes related to Ras signaling [KRAS, V-Raf-1 murine leukemia viral oncogene homolog 1 (RAF1), RAASF1] and cellular metabolism [ALPP, choline kinase alpha (CHKA), GGT1 and NAT2]. Hepatitis B had 133 genes shared with HCC and 12 with HCC-CC (24%). Genes shared between HBV and HCC-CC were alpha-fetoprotein (AFP), MET, KRT8, KRAS, RAF1, genes for metabolism and apoptosis. Similar to HBV, HCV had a high number of comparable genes when compared with HCC [115] and HCC-CC (9.18%). Similar to HBV, HCV demonstrated genes AFP, MET, KRAS and RAF1. Unlike HBV, HCV also demonstrated KRT18 (v. KRT8 for HBV), MMP7, TIMP3 and only GGT1 for metabolism.
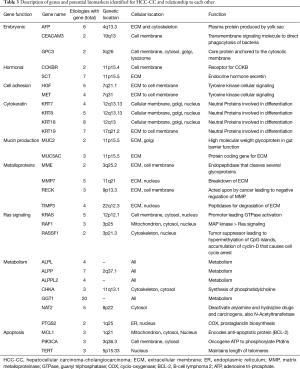
Full table
In addition to HCV, autoimmune disease such as AIH and PBC demonstrated several genes in common with HCC (88 for AIH and 42 PBC) and HCC-CC [5 (10%), 9 (18%) respectively]. AIH demonstrated GPC3, KRT8, 18, KRAS, GGT1 in common with HCC-CC. When comparing genes for PBC and HCC-CC, this etiology possessed SCT, HGF, KRT7, KRT19, MUC5AC, MMP7, ALPP, CHKA and myeloid cell leukemia 1 (MCL1). Fatty liver disease had 4 genes in common with HCC-CC and 41 with HCC.
Cholangiocarcinoma (CC) risk factors
When evaluating the genes present in CC, our analysis yielded 98 genes. A summary of CC-related risk factors is summarized in Table 2. The highest number of genes among CC risk factors included 97 for cystic disease of the liver, 58 for cholangitis, 43 for schistosomiasis and 41 for PSC. The lowest number of genes present was found in other congenital malformations of the bile ducts and gall bladder.
As in HCC, we then compared the CC risk factors with HCC-CC, depicted in Table 3. Among these etiologies, cystic disease of the liver demonstrated 17 genes in common with CC and 13 of 50 with HCC-CC (26%). Such genes included all cytokeratin-related genes along with AFP, CEACAM3, SCT, MME, Ras association (RalGDS/AF-6) domain family member (RASSF1) and NAT2. Biliary cysts, demonstrated 10 genes in common with CC and 8 (16%) with HCC-CC. These results can be summarized in Table S3.

Full table
The next most common etiology for HCC-CC from CC was cholangitis. This risk factor demonstrated 8 genes in common with CC and 7 (14%) with HCC-CC. This risk factor appeared to demonstrate several metabolic genes (ALPL, ALPP, ALPPL2 and GGT1), as well as KRT19 and MUC2. PSC demonstrated 2 (4%) genes in common with HCC-CC (KRT19 and GGT1) vs. 3 with CC alone. Choledochal cyst as a risk factor also demonstrated 2 (4%) genes in common with HCC-CC (SCT and PTGS2). Opisthochiasis, recurrent cholangitis, and other congenital malformations of the gall bladder demonstrated only a single gene in common with HCC-CC. All other CC-related etiologies had no risk factors in common with HCC-CC depicted in Table 3 and Table S3.
Discussion
Biphenotypic HCC-CC is a unique hepatic neoplasm expressing features of both HCC and CC. This tumor poses a diagnostic dilemma, utilizing radiologic imaging alone, and thus biomarkers would prove a valuable resource in this condition (23,24). To identify potential biomarkers that could assist clinicians in the diagnosis of this unique primary liver cancer, we utilized GeneCards®, the Human Gene Compendium.
Prior studies have validated the use of this database in the study of several medical conditions (27-33). Specifically, this compendium has assisted researchers in identifying genes vital to the prognosis and pathogenesis of multiple malignancies, as well as non-neoplastic liver diseases (30-33). A similar concept has been employed in the setting of HCC-CC in a recent abstract analyzed the genetic composition of 15 pathologic specimens of HCC-CC (34). These investigators descriptively identified genetic markers present in their specimens and compared them with HCC, CC and HCC-CC information.
Our study identifies KRAS, MET, PIK3CA and TP53 as potential biomarkers for HCC-CC. Each of these genes is involved in Ras-signaling, a process vital to oncogenesis. MET factor and HGF encode tyrosine kinases, which allow for further cell signaling from the ECM into the cytoplasm. Furthermore, these gene products also interact with PI3K, which via signaling cascades also activates Ras. Both MET and PIK3CA have been evaluated in prior study to help determine prognostic and chemotherapeutic information in non-small cell lung cancer, breast, gastric, among other cancers (35,36). The MET-HGF complex has been evaluated previously in HCC, CC and HCC-CC, the latter via a small study of 30 pathologic specimens (37). This study demonstrated excess expression of c-met solely in the CC portion of combined HCC-CC.
A similar specificity for the CC portion of biphenotypic HCC-CC has also been described in the literature with regard to KRAS (9,38). The KRAS oncogene has previously been suggested as a clinical biomarker for a variety of abdominal neoplasms, including HCC-CC (34,38). Cancers other than HCC-CC associated with differential expression of KRAS, include colorectal and pancreatic cancer. In colon cancer, KRAS expression has been shown to provide useful information regarding treatment strategies (39).
Biomarkers previously associated with HCC-CC were confirmed in this study and included: AFP, CEA, GGT1, cytokeratin, mucin and metalloproteinase genes (7-10,12-15,19). Among these biomarkers, GGT, AFP, and CEA have been non-specific in the setting of HCC-CC (25). In contrast, cytokeratin, mucin production, and metalloproteinase molecules appear to be valuable to the diagnosis and prognosis of HCC-CC (7-10,12-15,19,25). Specifically, they appear to be useful in differentiating between classical and stem cell subtypes of HCC-CC. Such determinations can be facilitated utilizing cytokeratin signaling biomarkers (7-10,12-15,19,25). These serologic markers also appear to be useful in colorectal and breast neoplasms, with similar application (40).
The overlap between HCC-CC and HCC’s pathologic subtypes was determined in our analysis to range between 0–24%, while CC subtypes demonstrated an overlap of 2–78%. Given the significantly larger genetic overlap between the subtypes of CC and HCC-CC, it would appear that HCC-CC is more likely a pathologic subtype of CC with features of HCC. Recently, overall survival for HCC and HCC-CC (41,42) has been assessed using the Surveillance, Epidemiology, and End Results (SEER) database. HCC-CC demonstrated an overall 1-, 3-, and 5-year survival rates of 26.5%, 12.4% and 9.2% (43). Additional analysis evaluating post-transplant prognosis documented a survival rate of 46% for HCC-CC as compared with 78% survival for HCC (44). When reviewing these results with prior studies of survival results for HCC and CC, the survival percentiles of HCC-CC are more consistent with CC (41-45). For CC, the survival after transplantation has been estimated at 22–42% of CC and 0-18% for CC without transplantation. The prognostic comparison and genetic overlap with CC suggests a shared pathogenesis for HCC-CC and CC.
Novel serologic markers identified in our analysis involve pathogenic roles in embryonic development, apoptosis and metabolism. The first of these unique genes, GPC3, codes for a cell surface heparin sulfate proteoglycan. This molecule inhibits the dipeptidyl peptidase activity of dipeptidyl peptidase-4 (DPP4), vital in apoptosis and growth regulation of several tissues. This gene has previously been utilized as a serologic marker for prognosis of HCC after curative resection (46). Down-regulation of this gene is correlated with uncontrolled cellular growth. Expression of this gene in the setting of HCC-CC has yet to be evaluated.
Our biomarker analysis also identifies were two genes involved in cellular metabolism. Choline kinase alpha (CHKA), encodes for enzymes that regulate the synthesis of phosphatidylcholine. The second gene, PTGS2, in combination with CHKA, is an additional metabolic target regulating biosynthesis of cyclo-oxygenase 2 (COX-2). The COX-2 enzyme is involved in inflammation and mitogenesis and has been implicated as a serologic marker for predicting the prognosis of prostate, breast and several other malignant conditions (47). Other biomarkers that may be valuable in obtaining prognostic information include two apoptosis genes, MCL1 and telomerase reverse transcriptase (TERT).
Over expression of telomerase reverse transcriptase (TERT) leads to cessation of telomere shortening, hence being associated with oncogenesis. Another apoptotic gene MCL1, encodes for an anti-apoptotic protein, which is a member of the Bcl-2 family. Alternate splicing of this protein leads to isoform1, which inhibits apoptosis directly. Both molecules appear to be useful in early detection of various malignancies, including HCC (48).
N-acetyltransferase 2 (NAT2) is also identified in our analysis. NAT2 has been implicated in the activation/inactivation of medications, as well as carcinogens. Subsequently, NAT2 may serve as a biomarker to predict the risk for drug induced liver injury (49). In prior studies of CC, genetic polymorphisms and upregulation of NAT2 have correlated with risk for CC (50). Such findings can be correlated with those listed above for KRAS and MET.
The culmination of the above genetic analysis demonstrates the utility of GeneCards in the analysis of low frequency malignancies such as HCC-CC. Not only did this genetic analysis confirm previously documented serologic markers for HCC-CC, it identifies several unique molecular targets, which may be useful in studies evaluating the pathogenesis, diagnosis, and prognosis of HCC-CC. It has also illuminated the similarity of HCC-CC with the pathologic subtypes of CC as compared to HCC. This genetic compendium also permits the creation of a map outlining relationships between several of these genes which may allow a better understanding of the pathogenesis of this rare primary neoplasm.
Despite these findings, potential weaknesses of this study include the retrospective evaluation of data collected from small numbers of patients. However utilizing a vast database, such as GeneCards®, The Human Gene Compendium, allows for an expanded evaluation of a rare disease. This approach has been validated in the past in similar neoplasms as well as more common liver disease. The ability to analyze a complex series of genetic components, which would be otherwise time and labor intensive, is an added benefit of this approach.
Although several novel cellular components and pathways have been identified as potential biomarkers for HCC-CC, the utility of each of these components or the combination of these biomarkers in clinical diagnosis and prognosis have yet to be determined. Nonetheless, employing large relational genetic databases such as GeneCards for an initial analysis will permit more focused investigation into the utility of biomarkers as well as guide studies of pathogenesis and future therapies.
Acknowledgements
None.
Footnote
Conflicts of Interest: AB Elfant is a consultant for Boston Scientific. The other authors have no conflicts of interest to declare.
References
- Wittekind C, Fischer HP, Ponchon T. Combined hepatocellular and cholangiocarcinoma. In: Hamilton SR, Aaltonen LA, editors. World Health Organization classification of tumours: pathology and genetics of tumours of the digestive system. Lyon: IARC Press, 2000:181-2.
- Park H, Choi KH, Choi SB, et al. Clinicopathological characteristics in combined hepatocellular-cholangiocarcinoma: a single center study in Korea. Yonsei Med J 2011;52:753-60. [Crossref] [PubMed]
- Sasaki M, Sato H, Kakuda Y, et al. Clinicopathological significance of 'subtypes with stem-cell feature' in combined hepatocellular-cholangiocarcinoma. Liver Int 2015;35:1024-35. [Crossref] [PubMed]
- Tang D, Nagano H, Nakamura M, et al. Clinical and pathological features of Allen's type C classification of resected combined hepatocellular and cholangiocarcinoma: a comparative study with hepatocellular carcinoma and cholangiocellular carcinoma. J Gastrointest Surg 2006;10:987-98. [Crossref] [PubMed]
- Yano Y, Yamamoto J, Kosuge T, et al. Combined hepatocellular and cholangiocarcinoma: a clinicopathologic study of 26 resected cases. Jpn J Clin Oncol 2003;33:283-7. [Crossref] [PubMed]
- Ng IO, Shek TW, Nicholls J, et al. Combined hepatocellular-cholangiocarcinoma: a clinicopathological study. J Gastroenterol Hepatol 1998;13:34-40. [Crossref] [PubMed]
- Hauben E, Struyf N, Michielsen P, et al. Cytokeratin profiles and mucin secretion in combined hepatocellular-cholangiocarcinoma. A case report. Pathol Res Pract 1996;192:488-91. [Crossref] [PubMed]
- Tickoo SK, Zee SY, Obiekwe S, et al. Combined hepatocellular-cholangiocarcinoma: a histopathologic, immunohistochemical, and in situ hybridization study. Am J Surg Pathol 2002;26:989-97. [Crossref] [PubMed]
- Coulouarn C, Cavard C, Rubbia-Brandt L, et al. Combined hepatocellular-cholangiocarcinomas exhibit progenitor features and activation of Wnt and TGFβ signaling pathways. Carcinogenesis 2012;33:1791-6. [Crossref] [PubMed]
- Itoyama M, Hata M, Yamanegi K, et al. Expression of both hepatocellular carcinoma and cholangiocarcinoma phenotypes in hepatocellular carcinoma and cholangiocarcinoma components in combined hepatocellular and cholangiocarcinoma. Med Mol Morphol 2012;45:7-13. [Crossref] [PubMed]
- Cazals-Hatem D, Rebouissou S, Bioulac-Sage P, et al. Clinical and molecular analysis of combined hepatocellular-cholangiocarcinomas. J Hepatol 2004;41:292-8. [Crossref] [PubMed]
- Chu KJ, Lu CD, Dong H, et al. Hepatitis B virus-related combined hepatocellular-cholangiocarcinoma: clinicopathological and prognostic analysis of 390 cases. Eur J Gastroenterol Hepatol 2014;26:192-9. [Crossref] [PubMed]
- Zhan Q, Shen BY, Deng XX, et al. Clinical and pathological analysis of 27 patients with combined hepatocellular-cholangiocarcinoma in an Asian center. J Hepatobiliary Pancreat Sci 2012;19:361-9. [Crossref] [PubMed]
- Mourad W, Khalaf H, Tulbah A, et al. Clinicopathologic features of hepatic neoplasms in explanted livers: a single institution experience. Ann Saudi Med 2007;27:437-41. [Crossref] [PubMed]
- Okuda H, Shiratori K, Yamamoto M, et al. Clinicopathologic features of patients with intrahepatic cholangiocarcinoma who are seropositive for alpha-fetoprotein-L3 and those with combined hepatocellular and cholangiocarcinoma. J Gastroenterol Hepatol 2006;21:869-73. [Crossref] [PubMed]
- Fujii H, Zhu XG, Matsumoto T, et al. Genetic classification of combined hepatocellular-cholangiocarcinoma. Hum Pathol 2000;31:1011-7. [Crossref] [PubMed]
- Koh KC, Lee H, Choi MS, et al. Clinicopathologic features and prognosis of combined hepatocellular cholangiocarcinoma. Am J Surg 2005;189:120-5. [Crossref] [PubMed]
- Jarnagin WR, Weber S, Tickoo SK, et al. Combined hepatocellular and cholangiocarcinoma: demographic, clinical, and prognostic factors. Cancer 2002;94:2040-6. [Crossref] [PubMed]
- Maeda T, Adachi E, Kajiyama K, et al. Combined hepatocellular and cholangiocarcinoma: proposed criteria according to cytokeratin expression and analysis of clinicopathologic features. Hum Pathol 1995;26:956-64. [Crossref] [PubMed]
- Tomimatsu M, Ishiguro N, Taniai M, et al. Hepatitis C virus antibody in patients with primary liver cancer (hepatocellular carcinoma, cholangiocarcinoma, and combined hepatocellular-cholangiocarcinoma) in Japan. Cancer 1993;72:683-8. [Crossref] [PubMed]
- European Association For The Study Of The Liver; European Organisation For Research And Treatment Of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 2012;56:908-43. [Crossref] [PubMed]
- Khan SA, Davidson BR, Goldin RD, et al. Guidelines for the diagnosis and treatment of cholangiocarcinoma: an update. Gut 2012;61:1657-69. [Crossref] [PubMed]
- Fowler KJ, Sheybani A, Parker RA 3rd, et al. Combined hepatocellular and cholangiocarcinoma (biphenotypic) tumors: imaging features and diagnostic accuracy of contrast-enhanced CT and MRI. AJR Am J Roentgenol 2013;201:332-9. [Crossref] [PubMed]
- Wells M, Venkatesh S, Johnson G, et al. Biphenotypic Intrahepatic Cholangiocarcinoma and Hepatocellular Carcinoma: Imaging Features on MRI, CT, Ultrasound and PET. Radiological Society of North America 2014 Scientific Assembly and Annual Meeting, Chicago IL. . Accessed January 4, 2015http://archive.rsna.org/2014/14007120.html
- O'Connor K, Walsh JC, Schaeffer DF. Combined hepatocellular-cholangiocarcinoma (cHCC-CC): a distinct entity. Ann Hepatol 2014;13:317-22. [PubMed]
- Wanless IR. International consensus on histologic diagnosis of early hepatocellular neoplasia. Hepatol Res 2007;37 Suppl 2:S139-41. [Crossref] [PubMed]
- Rappaport N, Twik M, Nativ N, et al. MalaCards: a comprehensive automatically-mined database of human diseases. Curr Protoc Bioinformatics 2014;47:1.24.1-19.
- Stelzer G, Dalah I, Stein TI, et al. In-silico human genomics with GeneCards. Hum Genomics 2011;5:709-17. [Crossref] [PubMed]
- Safran M, Dalah I, Alexander J, et al. GeneCards Version 3: the human gene integrator. Database (Oxford) 2010;2010:baq020.
- Kim JM, Kim HG, Han JM, et al. The herbal formula CGX ameliorates the expression of vascular endothelial growth factor in alcoholic liver fibrosis. J Ethnopharmacol 2013;150:892-900. [Crossref] [PubMed]
- Zhou T, Wang T, Garcia JG. Genes influenced by the non-muscle isoform of Myosin light chain kinase impact human cancer prognosis. PLoS One 2014;9:e94325. [Crossref] [PubMed]
- Sharma G, Agarwal SM. Identification of critical microRNA gene targets in cervical cancer using network properties. Microrna 2014;3:37-44. [Crossref] [PubMed]
- Baasiri RA, Glasser SR, Steffen DL, et al. The breast cancer gene database: a collaborative information resource. Oncogene 1999;18:7958-65. [Crossref] [PubMed]
- Keller JW, Doyle MM, Wang-Gilliam A, et al. Analysis of the genomic profile of biphenotypic tumors compared to cholangiocarcinoma and hepatocellular carcinoma. J Clin Oncol 2014;32:abstr 226.
- Santarpia M, Altavilla G, Margeli M, et al. PIK3CA mutations and BRCA1 expression in breast cancer: potential biomarkers for chemoresistance. Cancer Invest 2008;26:1044-51. [Crossref] [PubMed]
- Sierra JR, Tsao MS. c-MET as a potential therapeutic target and biomarker in cancer. Ther Adv Med Oncol 2011;3:S21-35. [Crossref] [PubMed]
- Varnholt H, Asayama Y, Aishima S, et al. C-met and hepatocyte growth factor expression in combined hepatocellular and cholangiocarcinoma. Oncol Rep 2002;9:35-41. [PubMed]
- Imai Y, Oda H, Arai M, et al. Mutational analysis of the p53 and K-ras genes and allelotype study of the Rb-1 gene for investigating the pathogenesis of combined hapatocellular-cholangiocellular carcinomas. Jpn J Cancer Res 1996;87:1056-62. [Crossref] [PubMed]
- Cavallini A, Valentini AM, Lippolis C, et al. KRAS genotyping as biomarker in colorectal cancer: a comparison of three commercial kits on histologic material. Anticancer Res 2010;30:5251-6. [PubMed]
- Ohlsson L, Israelsson A, Öberg Å, et al. Lymph node CEA and MUC2 mRNA as useful predictors of outcome in colorectal cancer. Int J Cancer 2012;130:1833-43. [Crossref] [PubMed]
- Nathan H, Pawlik TM, Wolfgang CL, et al. Trends in survival after surgery for cholangiocarcinoma: a 30-year population-based SEER database analysis. J Gastrointest Surg 2007;11:1488-96; discussion 1496-7. [Crossref] [PubMed]
- Weimann A, Varnholt H, Schlitt HJ, et al. Retrospective analysis of prognostic factors after liver resection and transplantation for cholangiocellular carcinoma. Br J Surg 2000;87:1182-7. [Crossref] [PubMed]
- Wang J, Wang F, Kessinger A. Outcome of combined hepatocellular and cholangiocarcinoma of the liver. J Oncol 2010;2010.
- Groeschl RT, Turaga KK, Gamblin TC. Transplantation versus resection for patients with combined hepatocellular carcinoma-cholangiocarcinoma. J Surg Oncol 2013;107:608-12. [Crossref] [PubMed]
- Nagorney DM, Kendrick ML. Hepatic resection in the treatment of hilar cholangiocarcinoma. Adv Surg 2006;40:159-71. [Crossref] [PubMed]
- Yang SL, Fang X, Huang ZZ, et al. Can serum glypican-3 be a biomarker for effective diagnosis of hepatocellular carcinoma? A meta-analysis of the literature. Dis Markers 2014;2014:127831.
- Wang D, Guo XZ, Li HY, et al. Prognostic significance of cyclooxygenase-2 protein in pancreatic cancer: a meta-analysis. Tumour Biol 2014;35:10301-7. [Crossref] [PubMed]
- El-Mazny A, Sayed M, Sharaf S. Human telomerase reverse transcriptase messenger RNA (TERT mRNA) as a tumour marker for early detection of hepatocellular carcinoma. Arab J Gastroenterol 2014;15:68-71. [Crossref] [PubMed]
- Higuchi N, Tahara N, Yanagihara K, et al. NAT2 6A, a haplotype of the N-acetyltransferase 2 gene, is an important biomarker for risk of anti-tuberculosis drug-induced hepatotoxicity in Japanese patients with tuberculosis. World J Gastroenterol 2007;13:6003-8. [Crossref] [PubMed]
- Prawan A, Kukongviriyapan V, Tassaneeyakul W, et al. Association between genetic polymorphisms of CYP1A2, arylamine N-acetyltransferase 1 and 2 and susceptibility to cholangiocarcinoma. Eur J Cancer Prev 2005;14:245-50. [Crossref] [PubMed]


